By Dato’ Dr Amar-Singh HSS, Consultant Paediatrician
As we implement our national COVID-19 vaccination programme, those of us who are healthcare professionals have numerous friends and acquaintances asking us about the safety of the COVID-19 vaccines. In particular is the issue of the risk of taking the vaccine if we have a history of allergies. It is recognised that some individuals get a rare, serious side effect, i.e. a severe allergic reaction called anaphylaxis. Anaphylaxis can occur after taking a drug, a bee sting, some food item, etc and not just vaccines; we often never know who is going to react in this way. This severe reaction may cause swelling of the face or body, breathing difficulties and a drop in the blood pressure. It is recognised that a few people have developed this after taking the COVID-19 vaccine. So we need to be informed about this and be able to discuss our risk with our doctor if we suffer from allergies.
As always it is important to look at the data and the evidence instead of relying on opinions or rumour. I have attempted to summarise our understanding about the risk of anaphylaxis from current available data.
How Common is Anaphylaxis after a COVID-19 Vaccine?
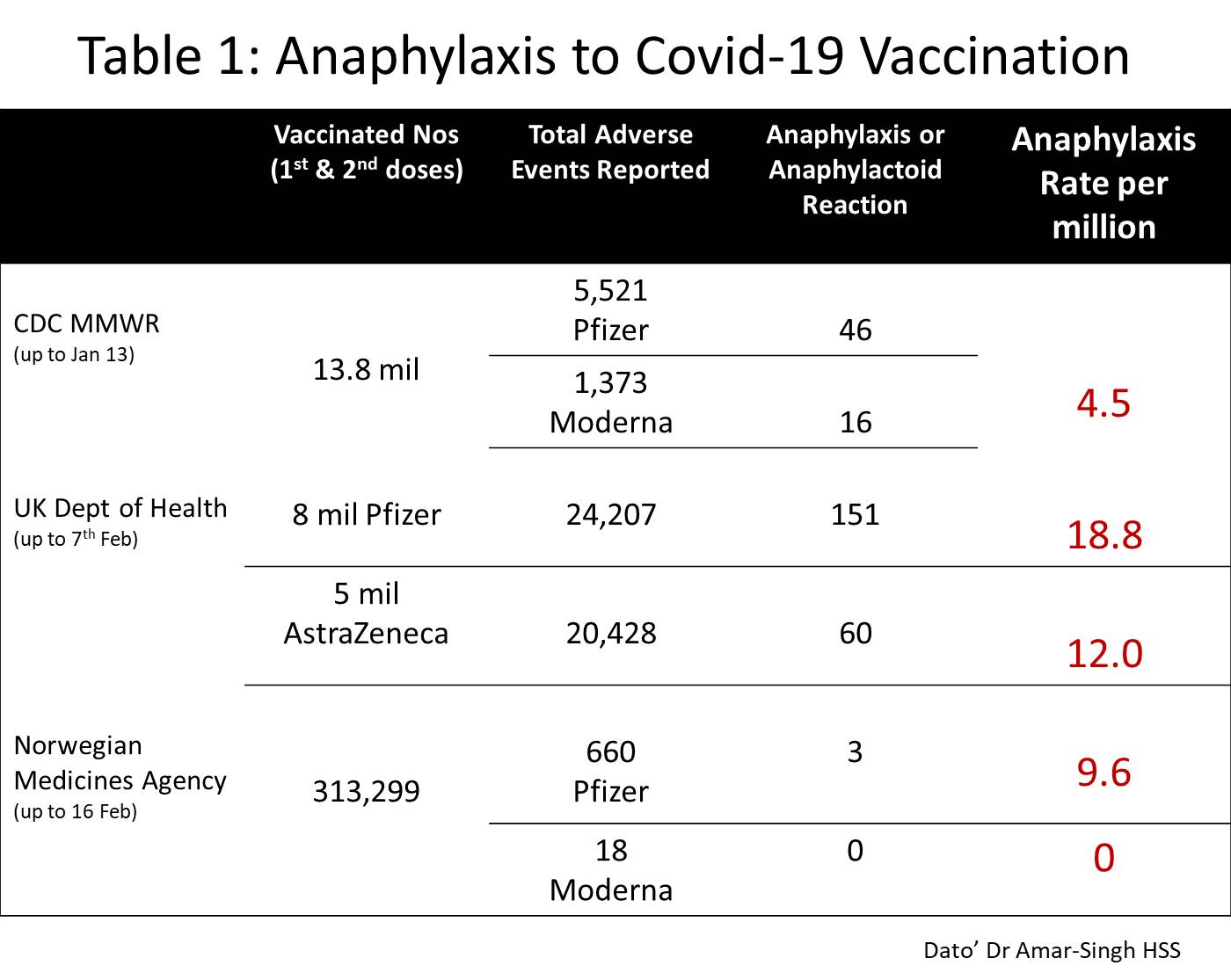
I have summarised the data from three different countries in Table 1 – the USA CDC Vaccine Adverse Event Reporting System, the UK Department of Health Yellow Card reporting system and the Norwegian ADR Registry of the Norwegian Medicines Agency.
These three countries report their experience of side effects. After more than 27 million doses, there were 276 confirmed anaphylaxis events, which gives a rate of 10.2 per million doses or ~1 per 100,000 persons vaccinated. There are different rates of anaphylaxis in different countries (different genetic background) and possibly different rates with different vaccines (need more data here).
This, however, means that anaphylaxis after COVID-19 is a very rare event. Note that all the people who experienced anaphylaxis were treated and discharged well.
What more Data do we Need?
Ideally we would like to see data that shows the risk of anaphylaxis in people who had prior anaphylaxis, and the risk of anaphylaxis in people with major or serious allergies (by type of vaccine). This means of all people vaccinated with serious allergies, how many actually developed anaphylaxis? The percentage (or rate) must be very, very small as many people have allergies.
The idea of this comes from a published US CDC Report that describes 21 people who developed anaphylaxis after taking the COVID-19 vaccine. 17 out of 21 (81%) had a documented past history of allergies or allergic reactions to drugs, medical products, foods and insect stings, and 7 out of 21 (33%) had experienced an episode of anaphylaxis in the past (1 after rabies vaccine; 1 after influenza A H1N1 vaccine).
Hence the risk of anaphylaxis is higher in those who have had a past history of allergies but occurs less often in those with no history of allergies. However, remember that a very large number of people with allergies have had the vaccine safely with no reactions.
What Constitutes a Severe Allergy?
Many people may have some allergy, often a mild one; it is estimated that 20-30% of the population have some allergic condition. Common mild allergic conditions would include eczema, allergic rhinitis, food allergy, urticaria, contact dermatitis, insect allergies, mild asthma, etc. Examples of moderate allergic conditions would be drug allergies and severe asthma especially when not controlled. Severe allergies would be a previous anaphylaxis, or prior angio-oedema (cutaneous oedema without blood pressure or respiration changes) or previous hypersensitivity reactions to vaccines or vaccine components (especially polyethylene glycol).
What do International Organisations Recommend?
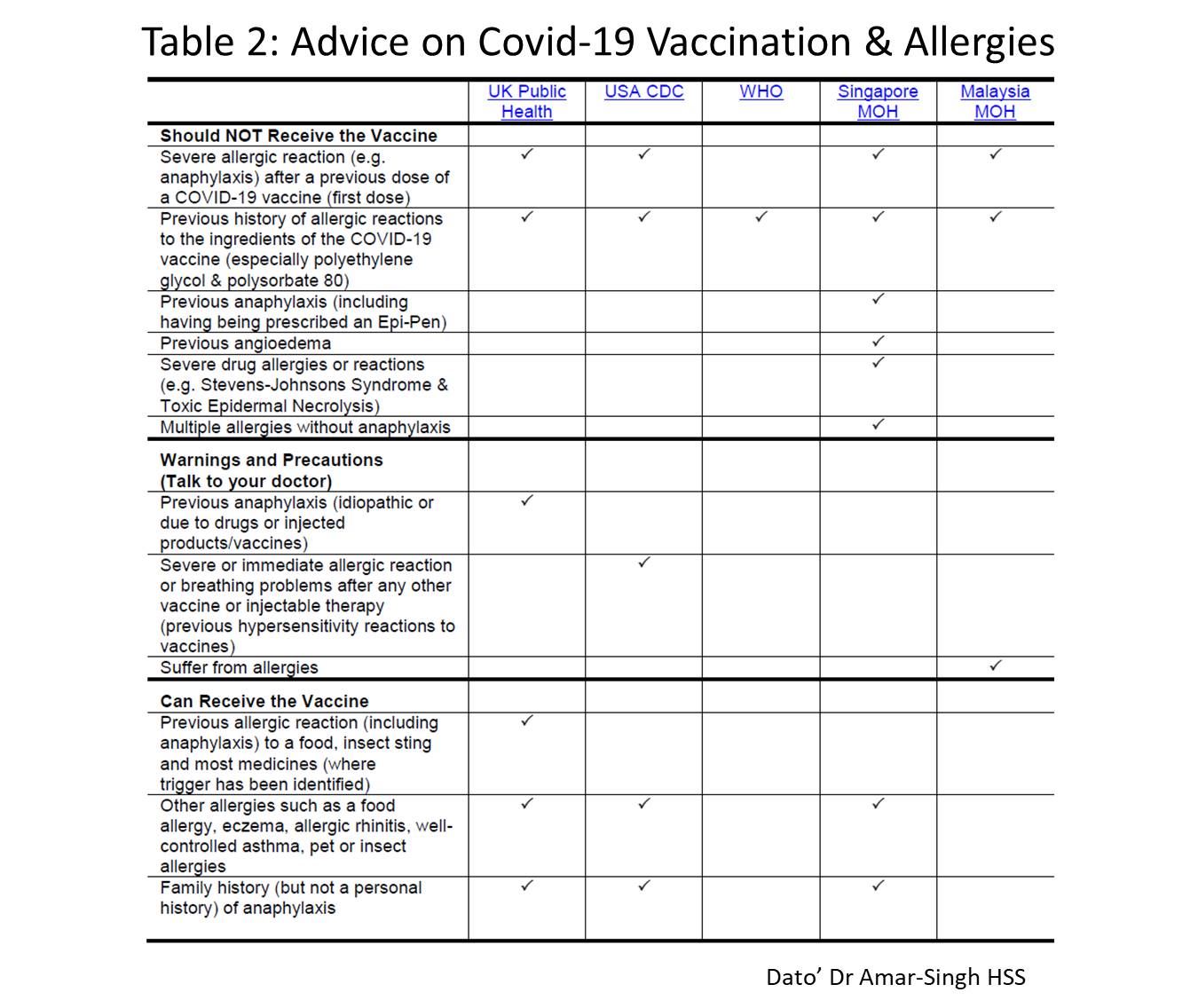
International organisations or regulatory bodies have made statements on COVID-19 vaccine use and allergies. Table 2 summaries their opinions.
Note that most support vaccination in those with prior allergies and some are even those who have had prior anaphylaxis (with close observation). But all do not support vaccination in anyone who has had a prior reaction to COVID-19 vaccines or a reaction to other vaccines or drugs that contained polyethylene glycol or polysorbate 80. Some are more cautious and conservative and hope for more data before removing some contraindications to vaccination.
What does our Ministry of Health (MOH) Advise?
The national COVID-19 immunisation programme booklet states that “The COVID-19 vaccine is safe for the majority of people. However, some groups will need further consideration prior to receiving the vaccine”. This includes “Individuals with severe allergies”. The National Pharmaceutical Regulatory Agency (NPRA) of MOH has a frequently asked questions (FAQ) section about the Pfizer (Comirnaty) vaccine. On allergies, they state that “Comirnaty should not be given to individuals who are known to have allergic reactions to any of the ingredients in the vaccine….The second dose of the vaccine should not be given to individuals who had a severe allergic reaction after the first dose of this vaccine.” They also advise individuals with allergies to speak to their doctor before deciding on their suitability for vaccination, but they also say that you may receive the vaccine even if you have an allergy. It would be good if clearer guidelines were made available to all doctors and health staff so they can advise those coming for vaccination. MOH will be monitoring all side effects after vaccination and encourage us to report them.
What do I do if I have Severe Allergies?
Having read all that, you may ask: What do I do if I am in the category of the person with a previous severe allergy (i.e. prior anaphylaxis or angio-oedema or a large number of bad drug allergies)? Well I am in the same situation as you are. I have numerous drug allergies, some very severe (Steven Johnson reaction); I have had an angio-oedema episode, one allergic airway obstruction and I also have underlying asthma and eczema.
We want to support the national COVID-19 vaccination programme, we want to help protect others by getting vaccinated and we certainly do not want our lives restricted if we don’t get vaccinated (if the government or businesses impose conditions on those not vaccinated).
I believe there are three possible options for those of us with severe allergies:
- Firstly we may choose not to get vaccinated as we feel the risk is too high. But we will have to accept a much stricter SOP for our lives and be prepared to have our life restricted for some time.
- The second option is that we choose to vaccinate and, due to the high risk, ask that it be done with enhanced health professional support, i.e. vaccinate in a hospital and have good physician backup with adrenaline at hand.
- The third option is to delay vaccination and watch the data as more is made available. The risk of anaphylaxis is small and likely to be even smaller as we get more vaccination data. We will also have a clearer idea as to which vaccine has the lowest rate of severe allergic reaction. Once this data is available, we hope to be able to choose the least allergic vaccine and vaccinate in a hospital with good physician backup.
Note that this advice only applies to those with severe allergies. Those with milder allergies should consider getting vaccinated but inform their doctor about their allergies. Hundreds of thousands of persons with allergies have already had the vaccine safely.
It would be good if the MySejahtera application, when registering for vaccination, had a clear box to check for past allergies. Currently we have only an ‘Others’ box to tick.
In this discussion we must remember that the chance of dying from COVID-19, especially if you are past 60 years like me, is much, much higher than any vaccine risk. Not to mention the 10-20% who may suffer from ‘long COVID’, which is quite debilitating. As always, we must appreciate that our vaccination will help protect those who cannot get vaccinated, especially children and those currently contraindicated due to poor health. What we need is full data transparency, especially about adverse events. Rapid, open granular data sharing with the public kills rumours and aids our vaccination programme.